Vela Diagnostics - Vela Diagnostics has announced that the ViroKey™ Flu A/B & RSV RT-PCR Extension Kit has received the CE mark for in vitro diagnostics use. “The flu season and the

Gerd Michel and Lou Welebob - Mutation Detection Simplified - From Liquid Biopsy in clinical oncolog - YouTube

Prospective Evaluation of the Vela Diagnostics Next-Generation Sequencing Platform for HIV-1 Genotypic Resistance Testing - ScienceDirect

Complete “Sample-to-Result” Highly Automated NGS and qPCR Workflow for Clinical Diagnostics Poster | Technology Networks

Vela Diagnostics - Vela Diagnostics has started shipping the ViroKey™ SARS-CoV-2 RT-PCR Test to laboratories and distributors across the #US, #Europe, #Thailand, #Vietnam, #Indonesia and #Dubai. Vela Diagnostics stands in solidarity with

La prueba ViroKey SARS-CoV-2 RT-PCR de Vela Diagnostics obtuvo la autorización de uso en emergencias de la FDA - COVID-19 - Hospimedica.es
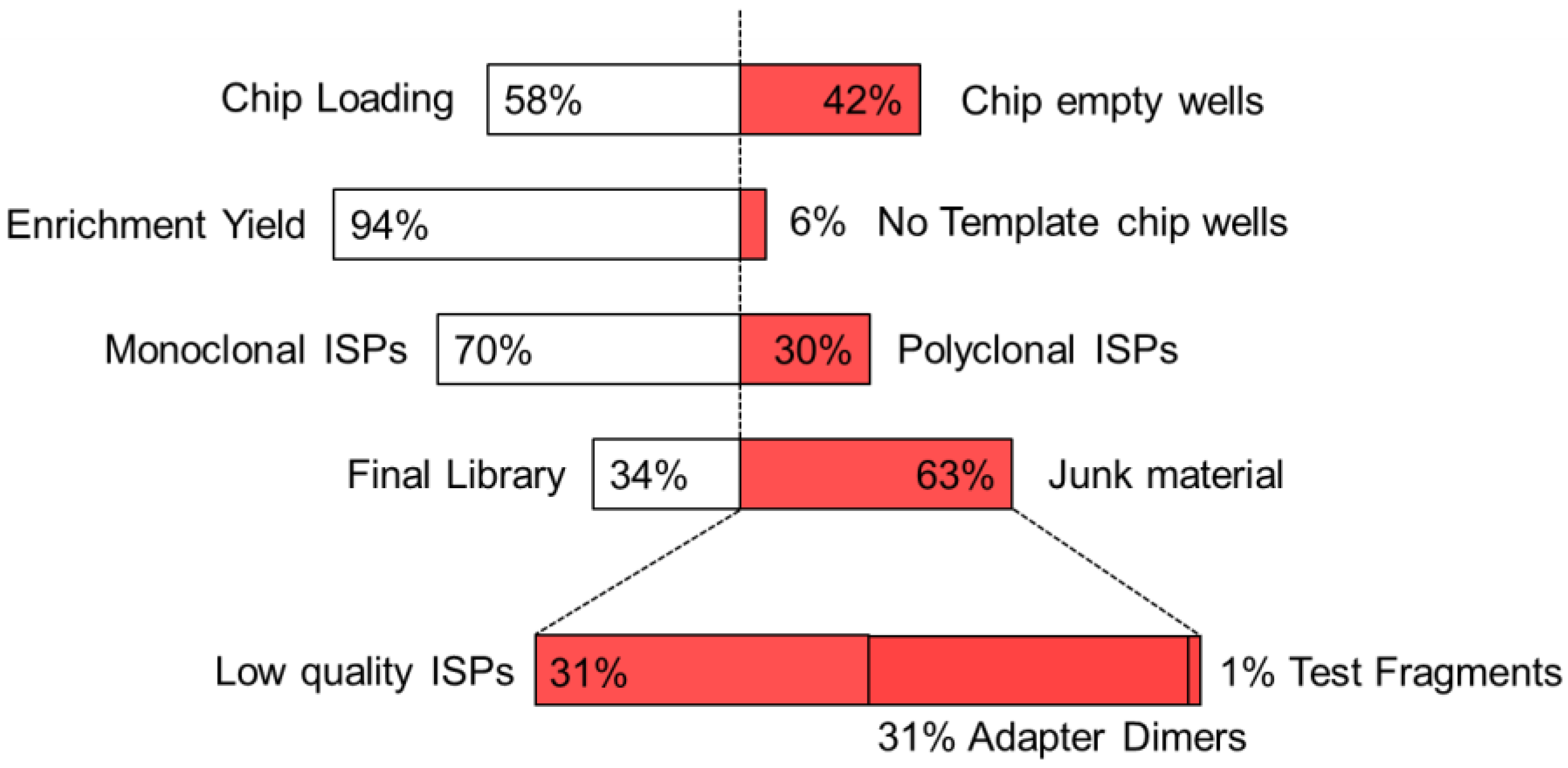
IJMS | Free Full-Text | Analytical Assessment of the Vela Diagnostics NGS Assay for HIV Genotyping and Resistance Testing: The Apulian Experience

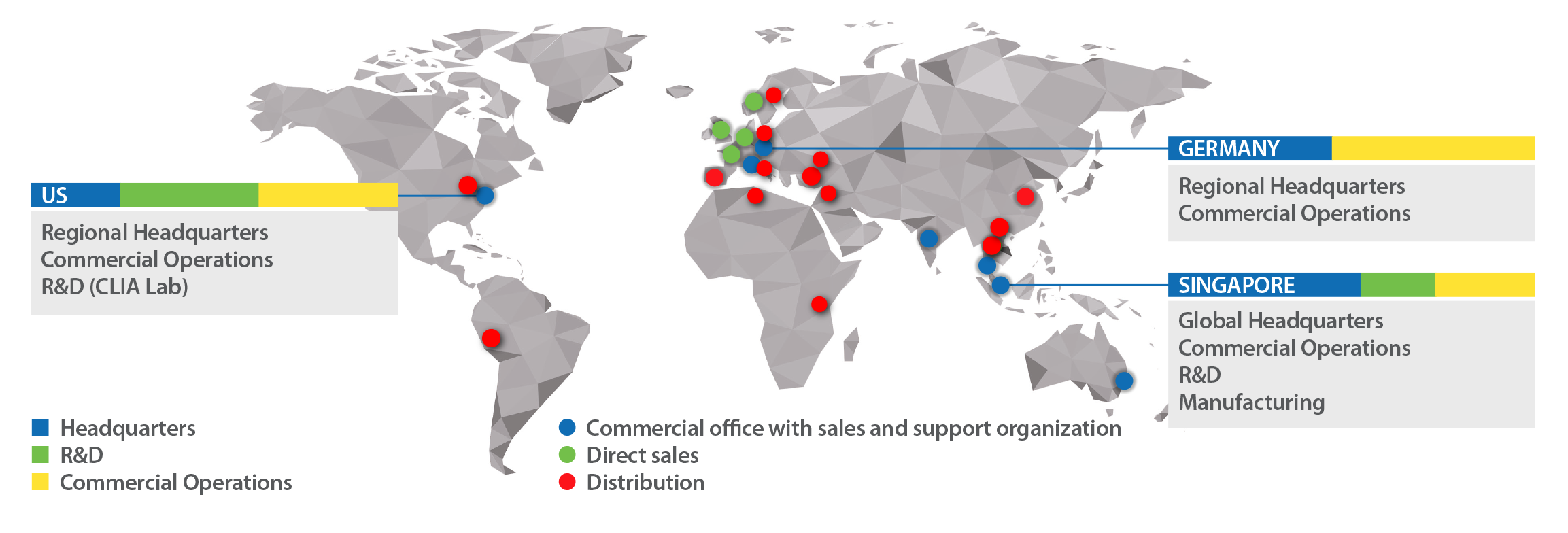
Global Provider of Integrated PCR & NGS Solutions with Automated Workflow and Data Reporting | Vela Diagnostics - Vela Diagnostics