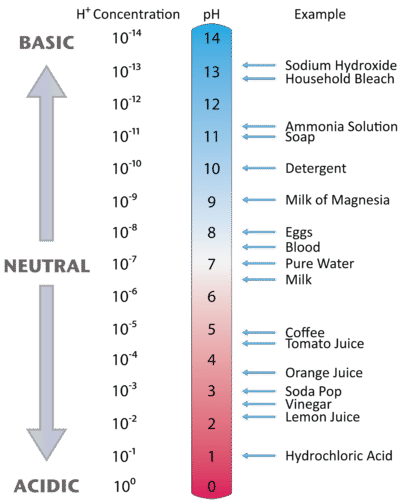
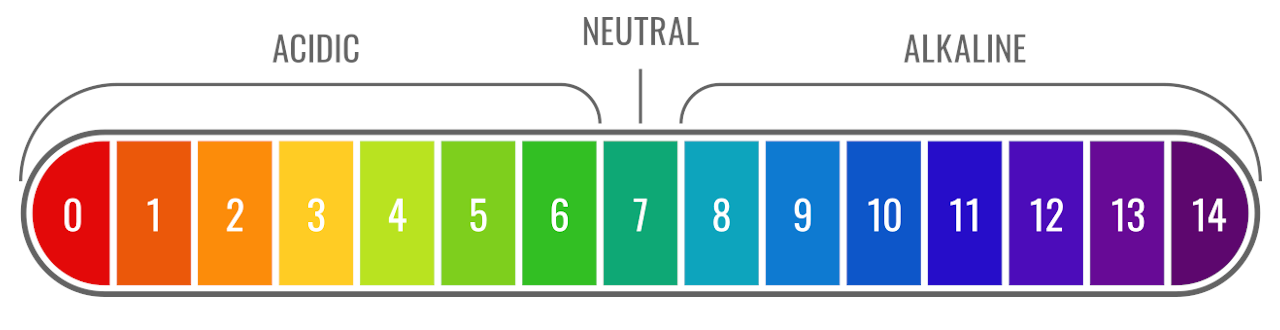
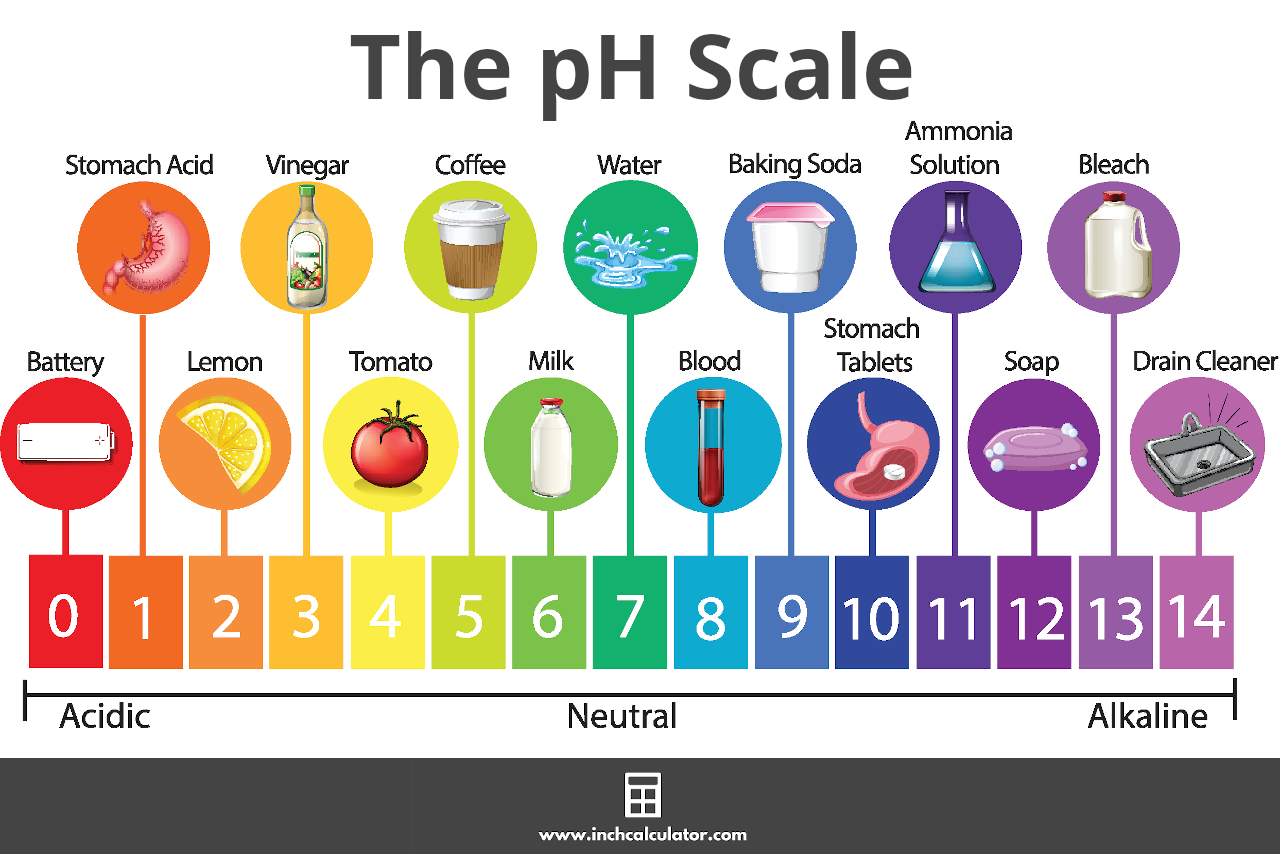
The pH of a solution describes its acidity and is the negative logarithm ( log) of its hydrogen ion concentration. The term pH is used because the hydrogen ion concentration in solutions of weak acids and in many other fluids is frequently much less than 1 ...

![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)











![pH = -log[H+] pH = -log[H+]](https://s3.studylib.net/store/data/008427242_1-6a856521ca6b3bcc442fa00c77f4fe6e.png)
![Given pH & pOH, Solve for [H+] & [OH-] Practice Problems - YouTube Given pH & pOH, Solve for [H+] & [OH-] Practice Problems - YouTube](https://i.ytimg.com/vi/Pm99tsEOyGQ/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AHCB4AC0AWKAgwIABABGGAgYChgMA8=&rs=AOn4CLBiQ7kUcjmLXRL3xjFxt985w-xdoQ)

![Calculate [H+] from pH - YouTube Calculate [H+] from pH - YouTube](https://i.ytimg.com/vi/yE4M2Uuh-h4/sddefault.jpg)