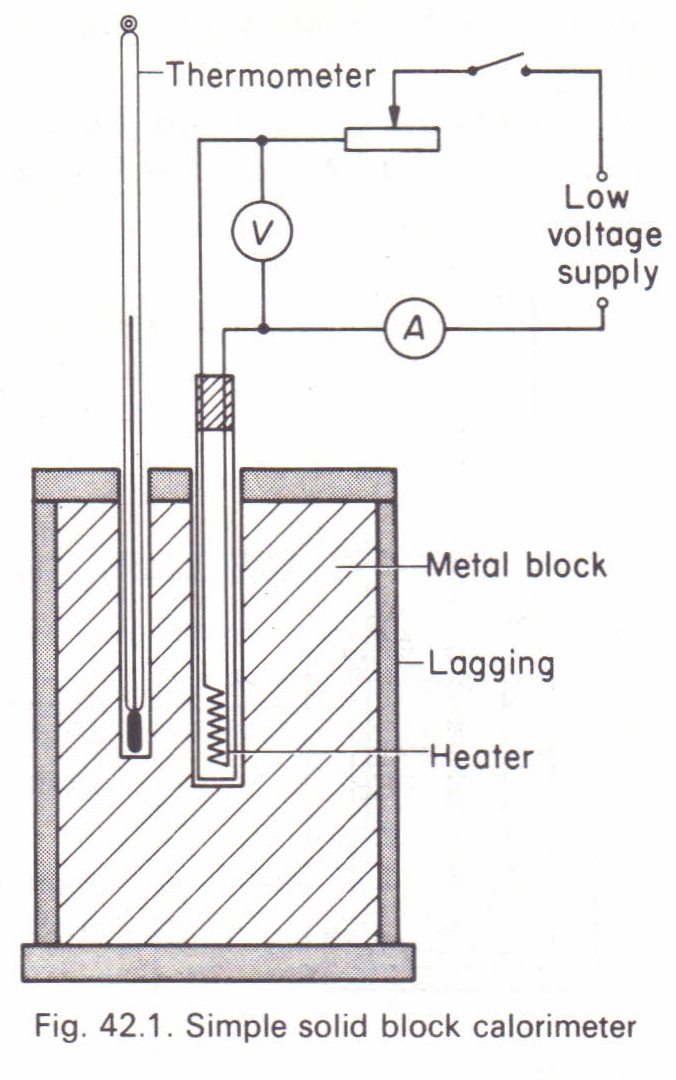
To measure the specific heat capacity of a metal (solid block method) Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

Cheat calculations Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

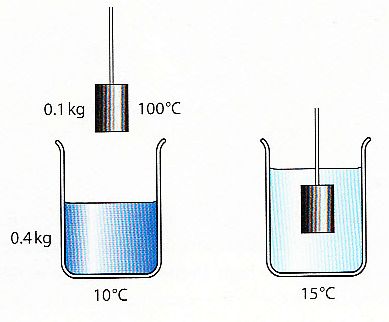
A mass of 50 g of a certain metal at 150^0C is immersed in 100 g of water at 11^0C. The final temperature is 20^0C . Calculate the specific heat capacity of

an experiment to calculate the specific heat capacity of a metal, the following results were obtained: - Brainly.in

A mass of 50 g of a certain metal at 150^@C is immersed in 100 g of water at 11^@C. The final temperature is 20^@C. Calculate the specific heat capacity of the




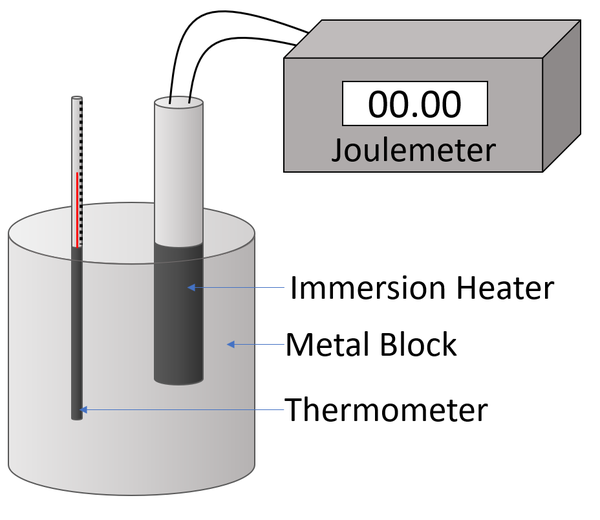



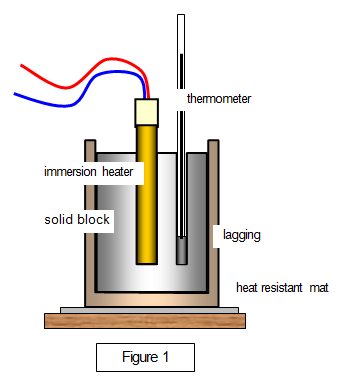




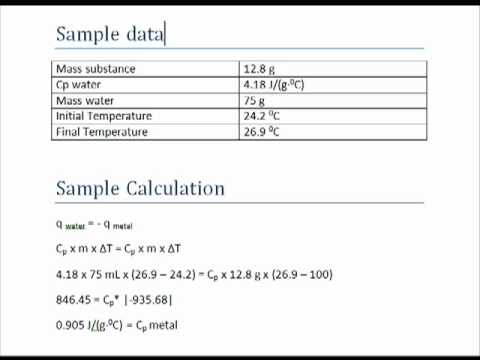





![Solved] Specific Heat Data of an unknown metal is | SolutionInn Solved] Specific Heat Data of an unknown metal is | SolutionInn](https://s3.amazonaws.com/si.experts.images/questions/2021/03/60501e6682161_image86.jpeg)