Vapor Pressure Formula & Example | How to Calculate Vapor Pressure - Video & Lesson Transcript | Study.com
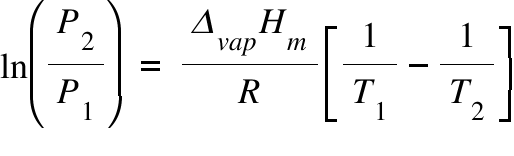
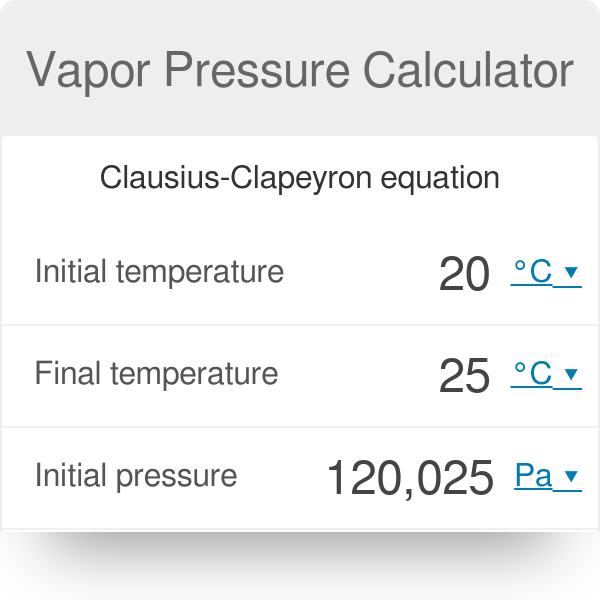
Correlation of Vapor Pressure at Different Temperatures by Clausius Clapeyron Equation Calculator | Calistry
Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.