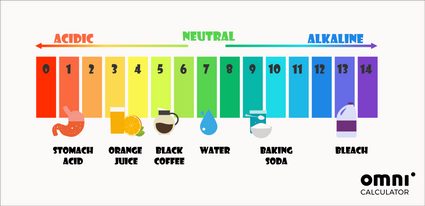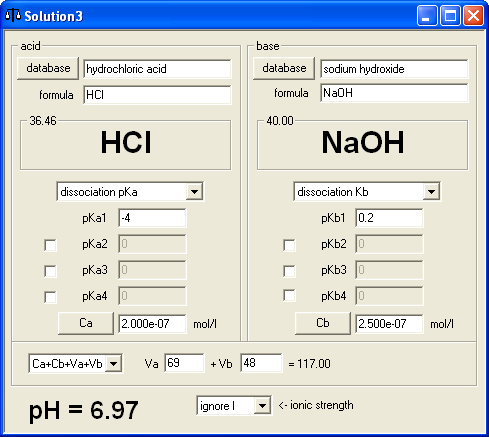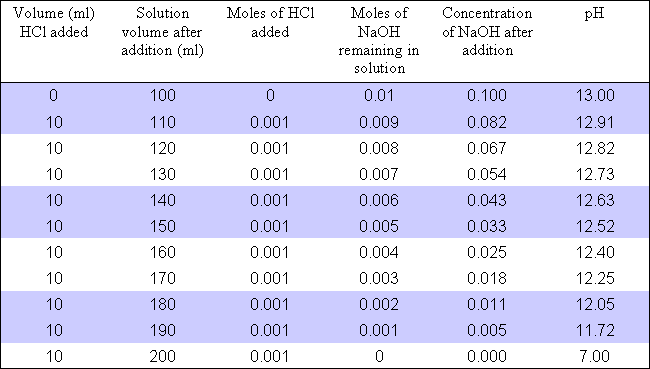
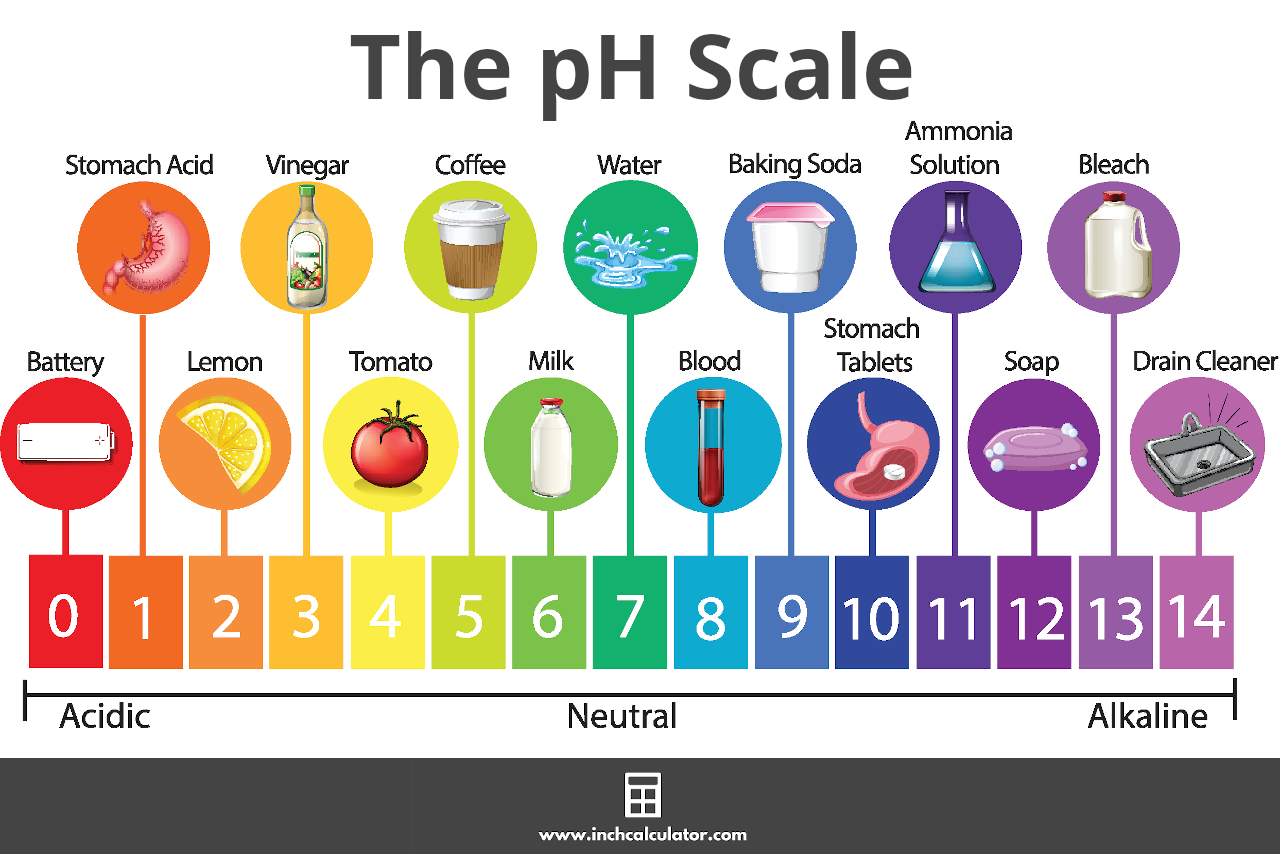
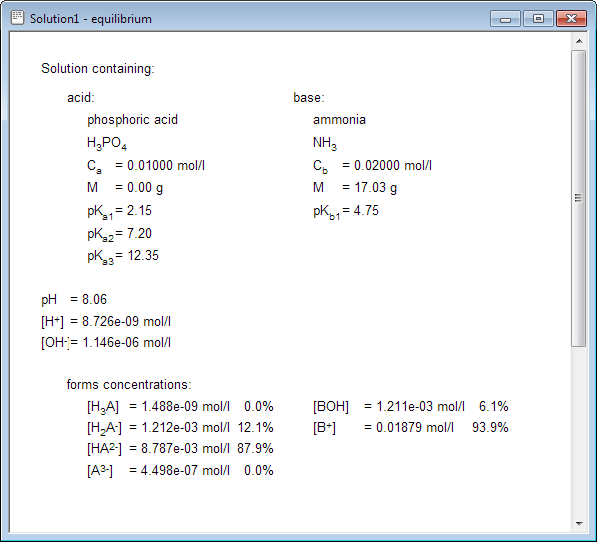
pH calculations and more in fundamentals of pharmaceutics. : Sodium hydroxide as a buffer in solution
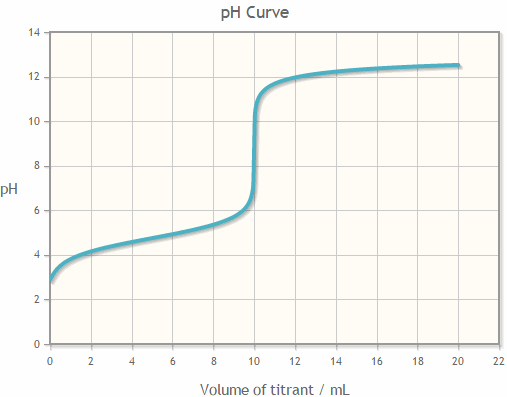
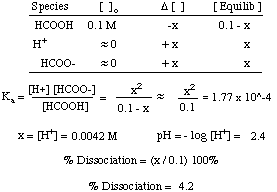
If an acetic acid solution is titrated with NaOH, how do I find out the amount of NaOH needed so the pH of the solution is exactly 7? Because the pH at
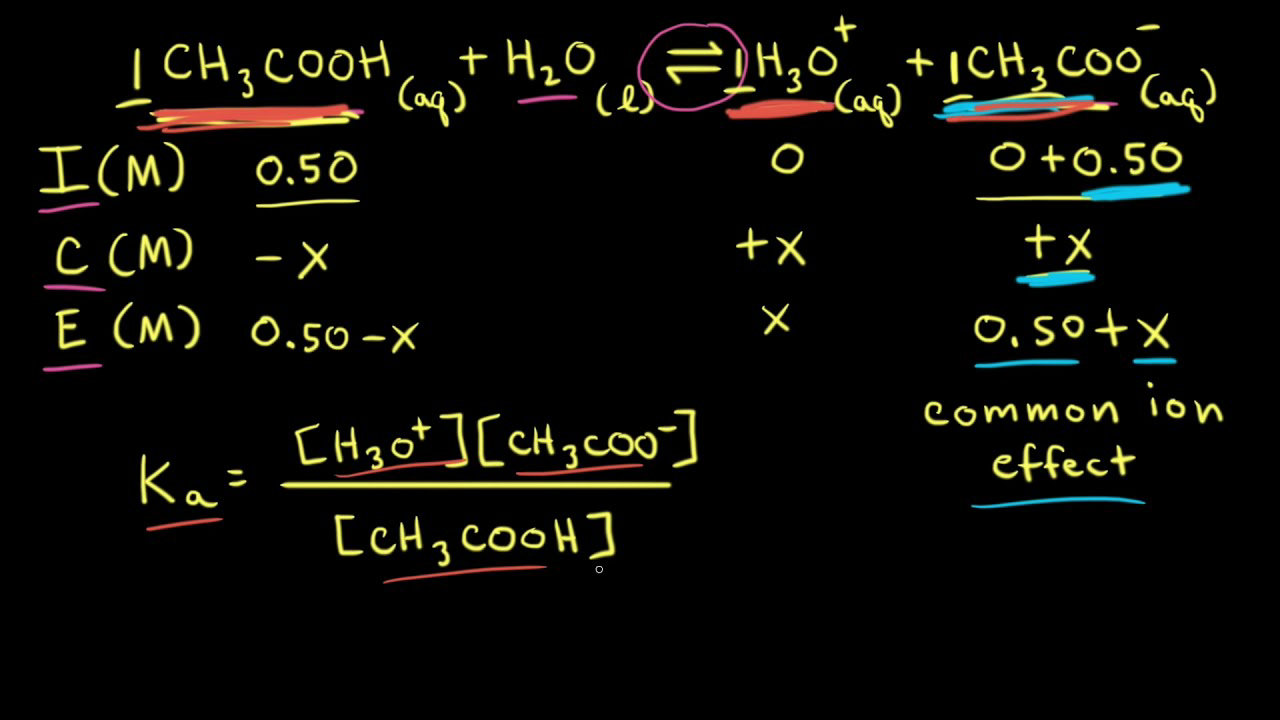





![Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/gn1CgBzShps/maxresdefault.jpg)
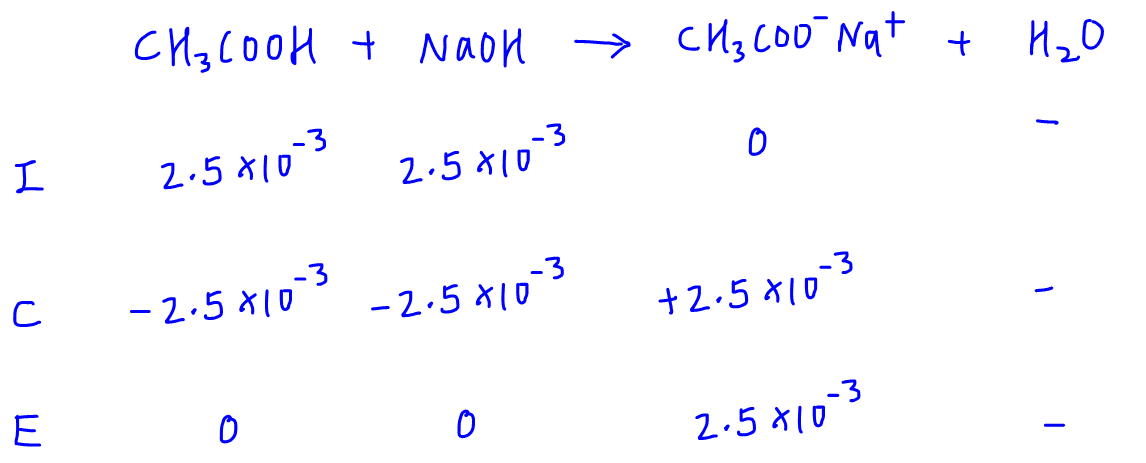


![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)