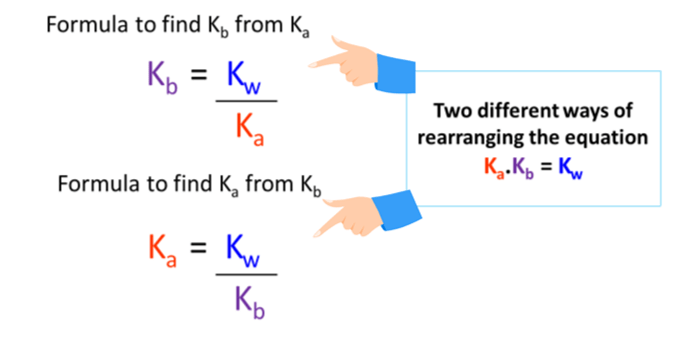
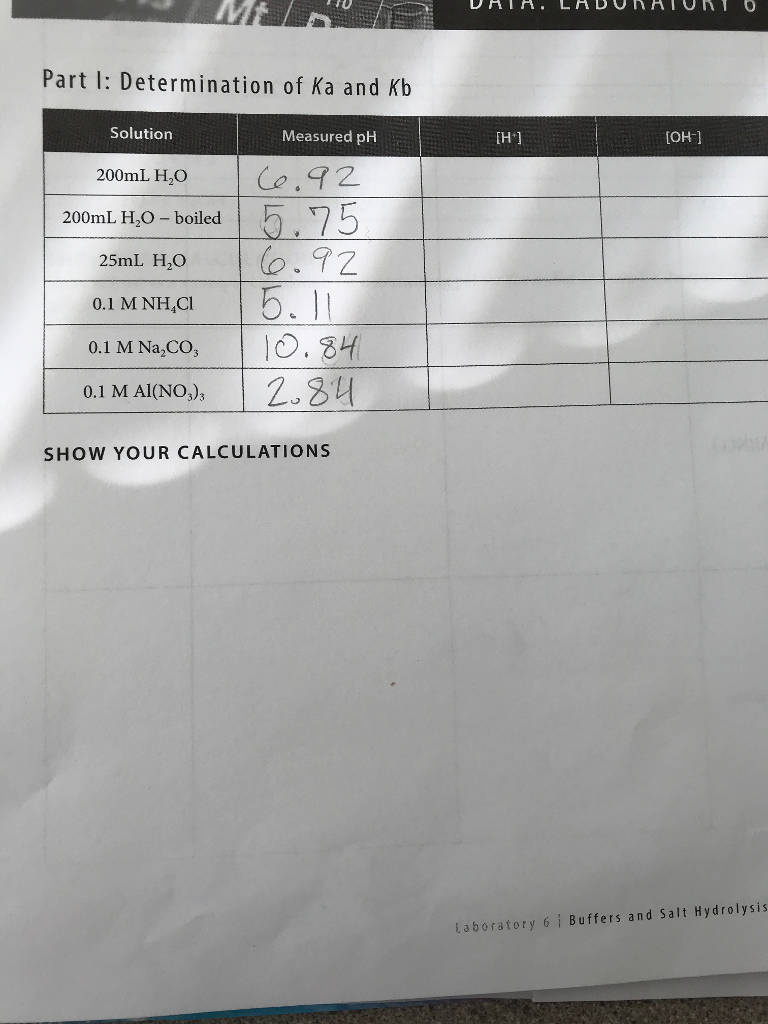
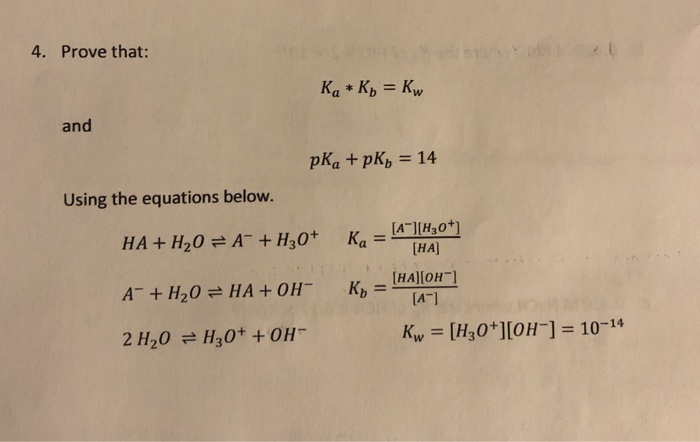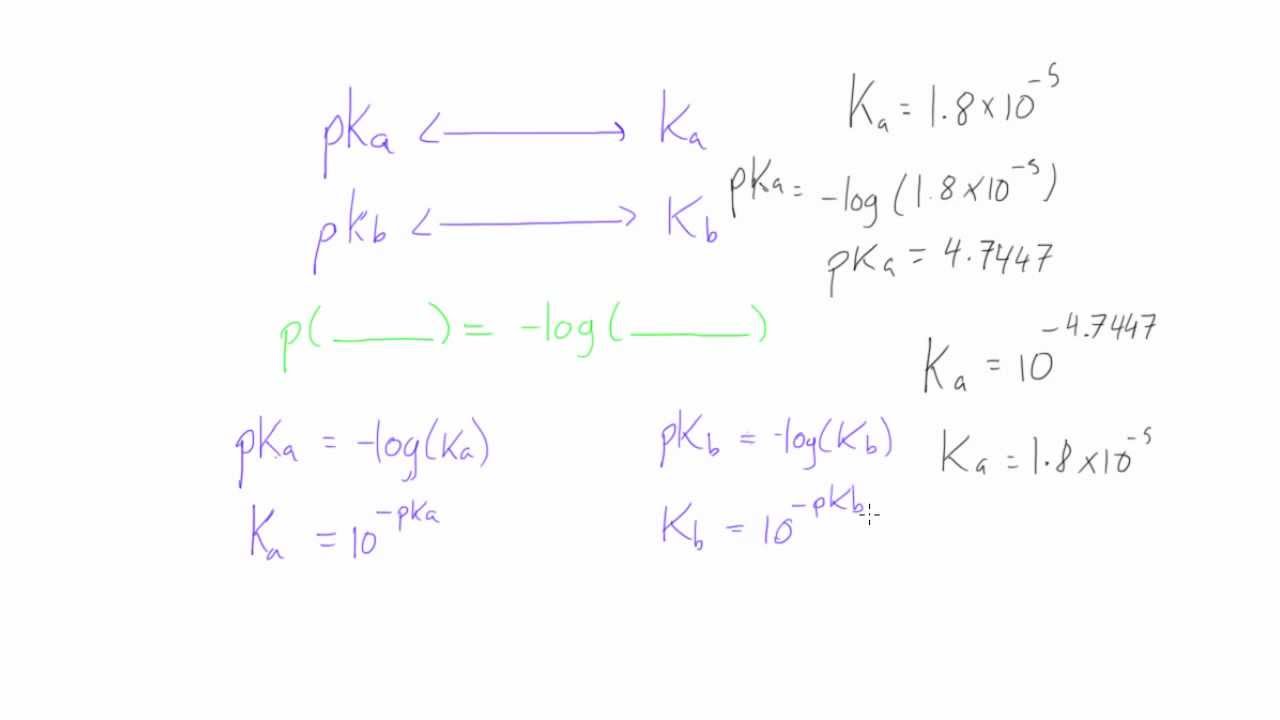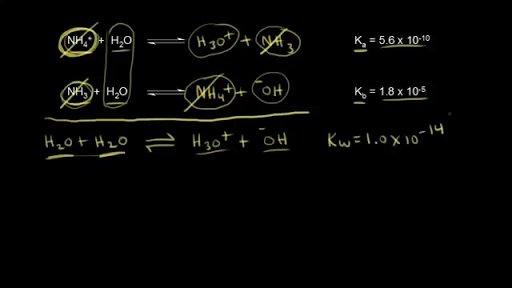The acid dissociation constant ka for an unknown acid ha is 4.57 x 10^-3 what is the base dissociation - Brainly.com

Acid-Base Equilibrium | Calculating the Ka or Kb of a Solution - Video & Lesson Transcript | Study.com

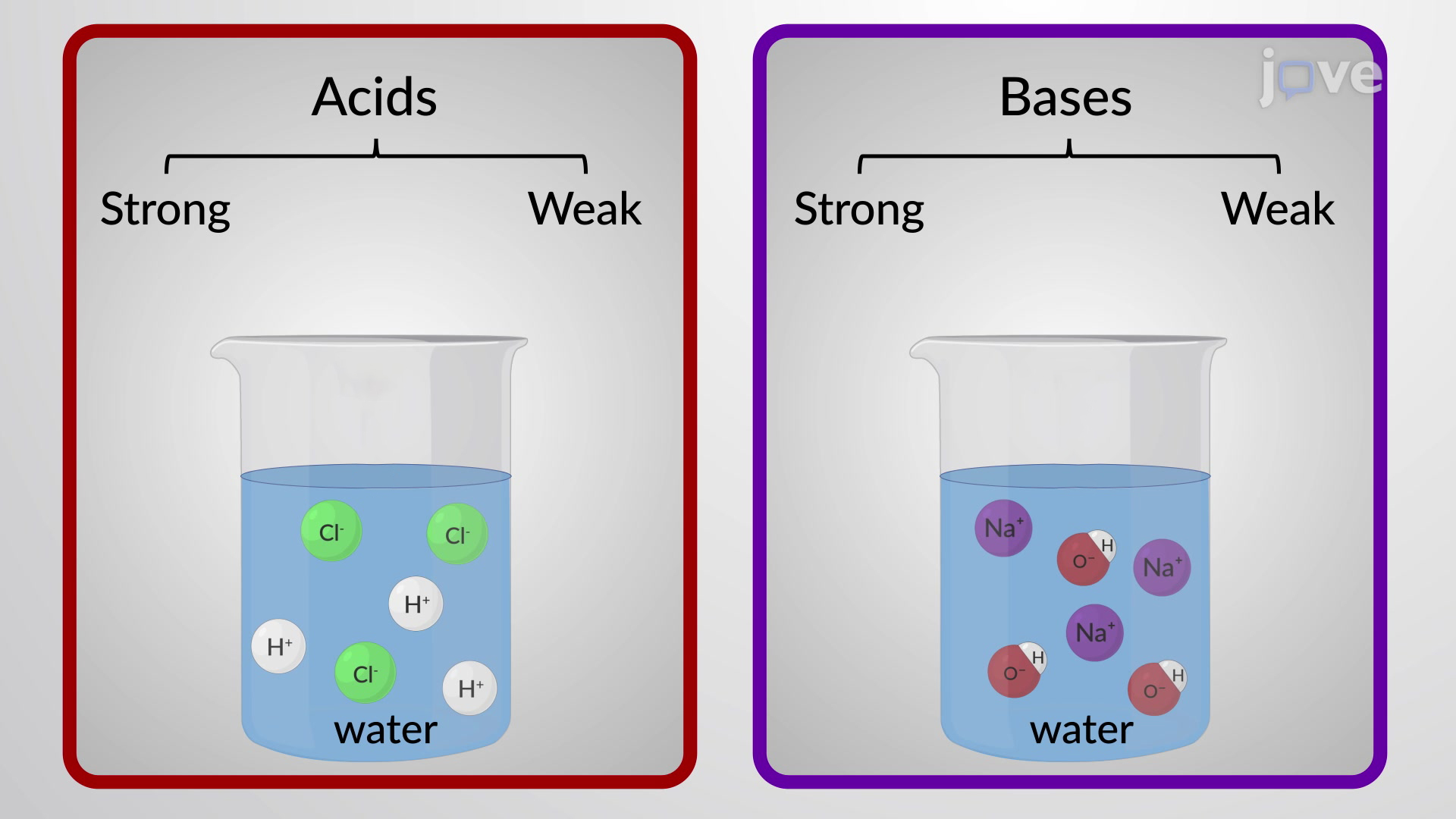
Calculate the pH of the following mixture given Ka = 1.8 × 10^-5 and Kb = 1.8 × 10^-5 ( pKa = pKa = 4.7447 ) 50mL 0.05M NaOH + 50mL of 0.1M CH3COOH