Calculate the value of Avogadro's number from the following data : Density of KF = 2.48 " g cm"^(-3) . Distance between K^(+) and F^(-) KF = 269 pm. (Atomic masses : K = 39 and F = 19 amu)

Calculate the value of Avogadro number from the internuclear distance of adjacent ions in NaCl , 0.282 nm and the density of solid NaCl is 2.17 × 10^3 kg/m^3 .A unit cell

Amazon.com: Holy Moley Avogadro Mole Calculator Chem Long Sleeve T-Shirt : Clothing, Shoes & Jewelry
a) Define: (i) Molecular mass, (ii) Avogadro constant. (b) Calculate the number of molecules in 50 g of CaCO3. (Atomic mass of Ca = 40 u, C = 12 u and O =

Casio fx-991EX calculator: HOW TO ACCESS CONSTANTS (Avogadro's constant, Planck's constant, etc.) - YouTube

Molar Volume: Using Avogadro's Law to Calculate the Quantity or Volume of a Gas - Video & Lesson Transcript | Study.com

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
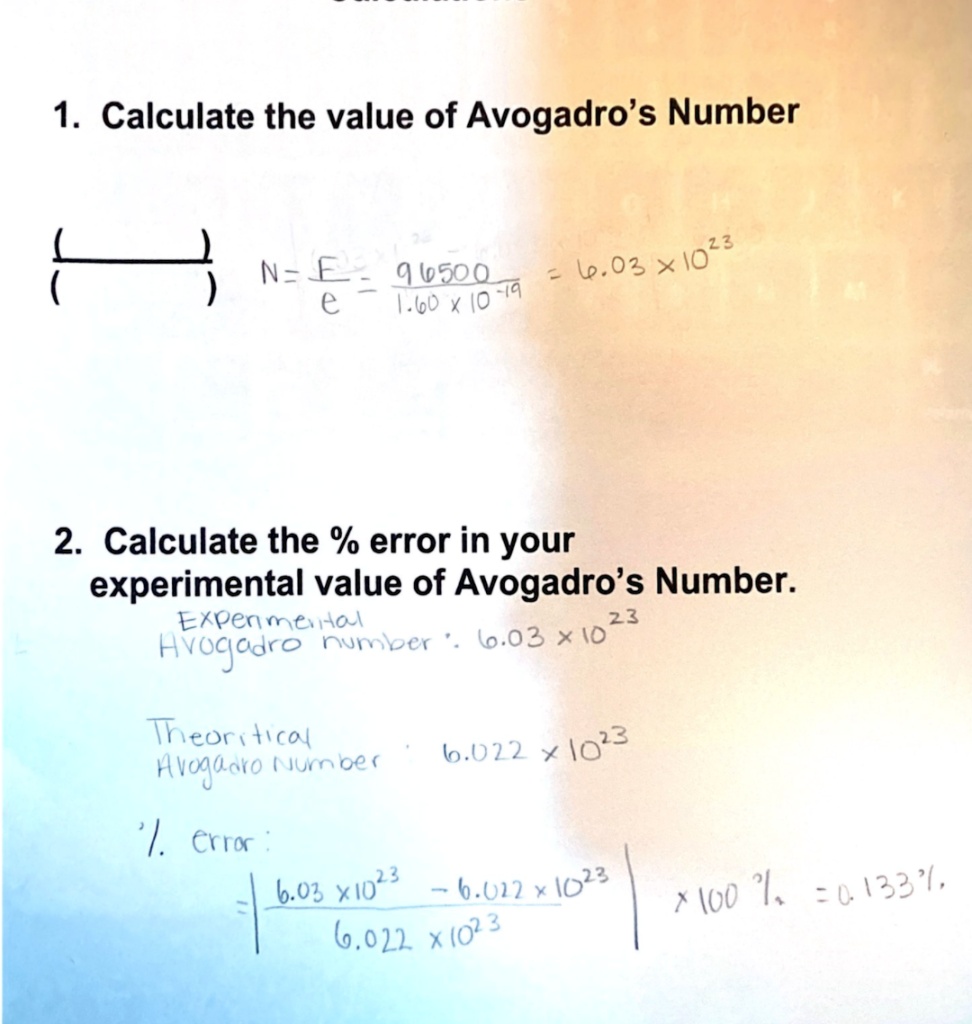
SOLVED: 1 Calculate the value of Avogadro's Number 13 l.03 x10 1 N= € 9.k5oo A.6o / I0 Fiq 2. Calculate the % error in your experimental value of Avogadro's Number: Ekpen





:max_bytes(150000):strip_icc()/Avogadro-58f7d6f35f9b581d5983024e.jpg)